Card payments are temporarily suspended for technical reasons! Please consider paying with cryptocurrencies with 15% off.
Card payments are temporarily suspended for technical reasons! Please consider paying with cryptocurrencies with 15% off.
days left





AUTHENTIC PRODUCTS
Only original pharmaceutical products that passed 3 stages of clinical trials.

GMP QUALITY
Our products are produced on GMP or state licensed factories.

10% CRYPTO DISCOUNT
10% discount for paying in BTC or other cryptocurrency.

PAYMENT BY CARD
Convenient Payment by Visa or MasterCard.

GUARANTEED DELIVERY
Guaranteed delivery times and clear Reshipment & Refund Rules.

PROMPT SHIPPING
Different shipping methods and same or next day shipping.
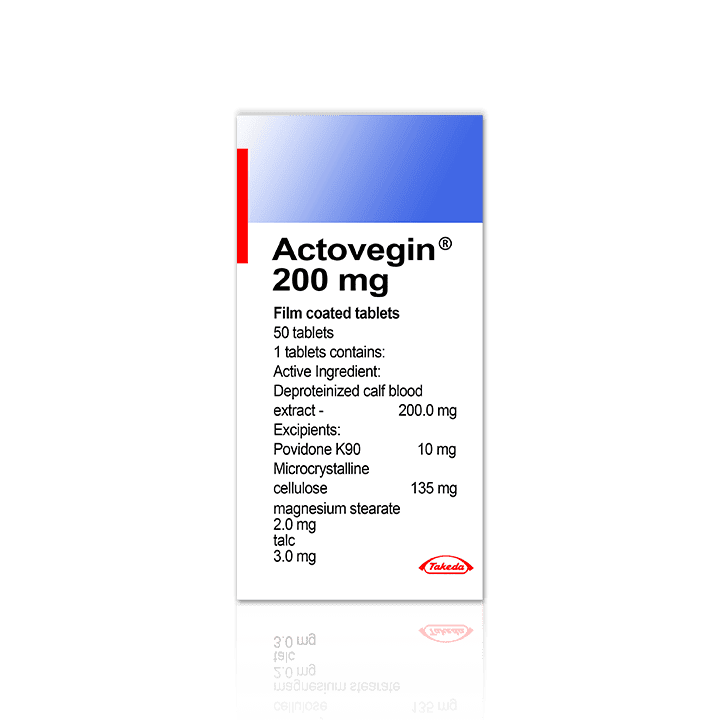
ACTOVEGIN ®
18 Reviews
Buy Actovegin® Online!
Actovegin is an ultra-filtered calf serum that enhances aerobic oxidation. It improves the absorption of glucose and oxygen uptake in tissue. That can provide a cell with more energy and potentially enhance its function.
| Chemical name | Actovegin |
| Form: | 5 ampoules of 5ml / 50 pills of 200mg |
| Form description: | Clear yellowish solution for injection / Glassy greenish-yellow film-coated tablets of a round biconvex shape |
| Active substance: | Actovegin (deproteinized hemoderivative of calf blood) |
| Excipients for pills: | Magnesium stearate – 2.0 mg, talc – 3.0 mg, povidone-K 90 – 10.0 mg, microcrystalline cellulose – 135.0 mg.; Coating: gum acacia – 6.8 mg, glycolic mineral wax – 0.1 mg, hypromellose phthalate – 29.45 mg, diethyl phthalate – 11.8 mg, quinoline yellow colorant – 2.0 mg, macrogol-6000 – 2.95 mg, povidone-K 30 – 1.54 mg, sucrose – 52.3 mg, talc – 42.2 mg, titanium dioxide – 0.86 mg |
| Excipients for vials: | Water for injection |
| СAS Number: | 63748-11-8 |
| ATC code: | B06AB |
| Pharmacological action: | Tissue regeneration stimulator |
| Shelf life: | 3 years. Do not use it beyond the expiration date printed on the package. |
| Storage conditions: | Store in a dry dark place at temperatures no higher than 25°C (77°F). Keep out of reach of children. |
| Terms of release: | Over-the-counter |
| Manufacturer: | Takeda Pharmaceutical, Austria/Russia https://www.takeda.com/ |
What is Actovegin?
Many decades ago shepherds in Switzerland noticed that young calves are able to heal their injuries and regenerate their tissue much faster than older calves. However, it was only in 1957 when Swiss scientists obtained a highly purified extract from the blood of young calves and developed the drug which is now, after countless improvements, called Actovegin.
Actovegin enhances aerobic oxidation, absorption of glucose, and oxygen uptake in tissue. For this reason, Actovegin has been used by athletes who wanted to speed up the recovery process and enhance physical performance and stamina. It is also deemed a great drug to treat various sports injuries.
As a starting material, the manufacturer uses the blood of calves under 3 months old. The extract is obtained by the ultrafiltration method, which ensures that there is no prion. There is also an Actovegin ointment with the same active ingredient which is sold under the brand name Solcoseryl.
What is Actovegin Composition?
The drug is a combination of various physiologically active ingredients:
- Amino acids, e.g. ornithine, leucine, alanine, glutamate, proline, glycine, taurine, etc;
- Oligopeptides;
- Lipids;
- Oligosaccharides;
- Nucleosides, e.g. adenosine, uridine;
- Antioxidants;
- Electrolytes, e.g. Na, Cl, K, Mg, Ca, F, Fe;
- Microelements, e.g. Cu, Si, Se, Zn.
The organic compounds account for 30% of the extract.
What are the Benefits and Effects of Actovegin?
Actovegin offers three kinds of effects: metabolic, neuroprotective, and microcirculatory.
- Actovegin increases the absorption and utilization of oxygen. It impacts positively on the utilization and transport of glucose, which leads to improved cellular metabolism and lowers lactate formation in case of ischemia.
- The neuroprotective effect of the drug is attributed to its ability to improve the regulation of apoptosis processes and inflammation in the central and peripheral nervous systems and repair single-chain DNA.
- The microcirculatory effect of Actovegin is due to an increase in the speed of capillary blood flow, a decrease in pericapillary space, a decrease in the myogenic tone of the precapillary arterioles and capillary sphincters, a decrease in the degree of blood flow with an improved circulation of blood in the capillary bed, as well as stimulation of endothelial nitric oxide synthase function, which influences the microcirculatory bed.
What is Actovegin Used For?
You can buy Actovegin for the following applications:
- Malfunction of the blood circulation and trophic disorders of the brain;
- Impairment of peripheral blood circulation and associated diseases;
- Cognitive impairments such as dementia and post-stroke cognitive impairments;
- Diabetic neuropathy as part of complex therapy;
- Dermal transplants and acute and chronic wounds.
Where Can I Order Actovegin Online?
You can order Actovegin online from different vendors. CosmicNootropic offers Actovegin in packs of 5x5ml ampoules and 50x200mg pills with worldwide shipping! Always be sure to do your research before purchasing any nootropic supplement to ensure you are getting a quality product.
As of 2015, Actovegin has been approved in 17 countries including Russia, China, South Korea, Latvia, and some others. You can order Actovegin and get it delivered to the USA, Europe, the UK, Asia, and many other destinations via CosmicNootropic.
Is Actovegin Banned by WADA?
Actovegin caused a lot of discussion and speculation after it was used by several athletes at the Sydney Olympic Games and Tour de France in 2000. The drug was suspected to have doping effects. However, the World Anti-Doping Agency (WADA) has not included Actovegin in its list of prohibited substances.
Some studies show that in order to induce considerable performance enhancement effects with Actovegin one has to exceed the permitted injectable amount of 50 ml every 6 hours.
Legal Disclaimer
This product has not been approved by the US FDA. All statements on this page are for informational purposes only and have not been evaluated by the US FDA.
This product is not intended to diagnose, treat, cure, or prevent any disease. See more
What is Actovegin Dosage and Administration?
Actovegin Pills
1-2 tablets 3 times per day before meals. Do not chew the tablet. Drink some water after the administration. The course duration is 4–6 weeks.
Actovegin Injections
5–10 ml intramuscularly or intravenously 3-4 times a week.
In case of mild metabolic disturbances – the dose is 5 ml. In case of severe metabolic disturbances, traumatic brain injury, and strokes the recommended dose of Actovegin solution is 10 ml.
What are the Side Effects of Actovegin?
As a rule, the compounds of Actovegin are very well tolerated. However, depending on the ROA and the dose, the drug can cause allergic reactions.
Can I Overdose on Actovegin?
Overdose cases have not been reported.
OFFICIAL INSTRUCTION
- Baiandin et al (1992) The effect of trental and actovegin on the organs of vision when administered into the end periorbital branches of the ophthalmic artery (experimental research) https://www.ncbi.nlm.nih.gov/pubmed/1295186
- Ziegler et al (2009) Treatment of symptomatic polyneuropathy with actovegin in type 2 diabetic patients https://www.ncbi.nlm.nih.gov/pubmed/19470838
- Elmlinger et al (2011) Neuroprotective and anti-oxidative effects of the hemodialysate actovegin on primary rat neurons in vitro https://www.ncbi.nlm.nih.gov/pubmed/21983748
- Lee et al (2011) Our experience on Actovegin, is it cutting edge? https://pubmed.ncbi.nlm.nih.gov/21271496/
- Machicao et al (2012) Pleiotropic neuroprotective and metabolic effects of Actovegin’s mode of action https://www.ncbi.nlm.nih.gov/pubmed/22910148
- Dieckmann et al (2012) Treatment with Actovegin® improves sensory nerve function and pathology in streptozotocin-diabetic rats via mechanisms involving inhibition of PARP activation https://www.ncbi.nlm.nih.gov/pubmed/22020669
- Guekht et al (2013) A Randomised, Double-Blind, Placebo-Controlled Trial of Actovegin in Patients with Post-Stroke Cognitive Impairment: ARTEMIDA Study Design https://www.ncbi.nlm.nih.gov/pubmed/24516413
- Melin et al (2014) Treatment with Actovegin improves spatial learning and memory in rats following transient forebrain ischaemia https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4190908/
- A Fomin, D Pershakov (2015) Actovegin in treatment of critical limb ischemia https://www.ncbi.nlm.nih.gov/pubmed/26356060
- Trofimova et al (2016) An experimental in vitro study of antioxidant and antiradical properties of cytoflavin, vinpocetine, actovegin and ethylmethylhydroxypyridine succinate https://www.ncbi.nlm.nih.gov/pubmed/27500876
- Søndergård et al (2016) Actovegin, a non-prohibited drug increases oxidative capacity in human skeletal muscle https://pubmed.ncbi.nlm.nih.gov/26744809/
- Guekht et al (2017) ARTEMIDA Trial (A Randomized Trial of Efficacy, 12 Months International Double-Blind Actovegin): A Randomized Controlled Trial to Assess the Efficacy of Actovegin in Poststroke Cognitive Impairment https://www.ncbi.nlm.nih.gov/pubmed/28432265
- Y Vakhrushev, M Lyapina (2017) Correction of small bowel function as a new direction for treating patients with metabolic syndrome https://www.ncbi.nlm.nih.gov/pubmed/28914850
- Ziegler et al (2017) Predictors of response to treatment with actovegin for 6 months in patients with type 2 diabetes and symptomatic polyneuropathy https://www.ncbi.nlm.nih.gov/pubmed/28438471
- Brock et al (2018) Update on the Role of Actovegin in Musculoskeletal Medicine: A Review of the Past 10 Years https://www.ncbi.nlm.nih.gov/pubmed/29337720
- Safarova et al (2018) Methods of augmentation of antidepressant therapy (on the model of complex therapy with the inclusion of actovegin) in gerontopsychiatric hospital https://www.ncbi.nlm.nih.gov/pubmed/30346435
- Firan et al (2020) Current Synthesis and Systematic Review of Main Effects of Calf Blood Deproteinized Medicine (Actovegin®) in Ischemic Stroke https://pubmed.ncbi.nlm.nih.gov/32365943/
- Li et al (2021) Neuroprotective Effects of Deproteinized Calf Serum in Ischemic Stroke https://pubmed.ncbi.nlm.nih.gov/34557139/
- La Fleur et al (2022) Actovegin in the management of patients after ischemic stroke: A systematic review https://pubmed.ncbi.nlm.nih.gov/35771887/
5
Based on 18 Reviews
(18)
(0)
(0)
(0)
(0)
04/02/2024
Violet E.
Verified Buyer
US
Really has Helped with an Unexpected Pleasant Surprise
I purchased this to help prevent cognitive aging. I'm really nervous about developing dementia that runs in my family. I've been struggling to focus and thinking clearly in my stressful job. This has helped lift the brain fog and reduce my stress level due to being about to think more clearly and resolve issues more quickly. I've also noticed the anti-inflammatory effect when injecting in my leg muscle that has sciatic pain. That's such an amazing unexpected effect! I'll definitely be purchasing this again!
04/02/2024
Dear Violet, we are so happy to hear about your positive experience with this product! Thank you for sharing with us :)
01/22/2024
Nicholas C.
Verified Buyer
US
Great product for reducing inflammation
Bought it for my wife who has severe osteoarthritis in her hands and it has helped quite a bit
01/26/2024
Dear Nicholas, thank you for your review, we are glad the product was helpful for your wife. We wish the health of your family!
01/09/2024
Eric A.
Verified Buyer
US
Actovegin - The Real Deal
I was skeptical but my cardio endurance went through the roof with Actovegin. I hope they restock soon!
01/09/2024
Dear Eric, thank you for your feedback, we are glad you are satisfied with the product!
12/29/2023
Anil R.
Verified Buyer
MY
Actovegin - IV for anti-aging
Using it monthly and feeling GREAT - other people comment as well....hehe
12/29/2023
Hi Anil! We are so happy to hear that you are satisfied with this product :) Thank you for leaving the review!
12/19/2023
jaime m.
Verified BuyerGreat quality
Since using actovegin my max heart rate has gone up 2 bpm and my treshold heart rate has gone 1 bpm. Since i started using it have a faster heart rate recovery and a lesser heart rate with the same effort as before.
12/22/2023
Dear Jaime, thank you for sharing your experience, we really appreciate it!
09/23/2023
Anil R.
Verified Buyer
MY
Actovegin - IV for anti-aging
No adverse reaction.....taking it IV in saline solution with other vitamins....ALL GOOD....let me keep trying for a few more months, will re-order for wife's stock
10/08/2023
Dear Anil, we're thrilled to know you liked the product. Please feel free to share your final thoughts on Actovegin after a course!
09/03/2022
formosa p.
Verified Buyer
US
good stuff very strong
good stuff very strong give alot energy
11/23/2021
Gerrard H.
Verified Buyer
CA
Wow
It’s amazing it heals all my injuries 25 % faster
09/09/2021
Mieszko S.
Verified Buyer
PL
Perfect preparation
It works very well. Intravenous or muscle application works and works as it should. In my case, the inflammation of the shoulder tendons is relieved and healed after a month of treatment.
04/05/2020
Timmo
Verified BuyerPerfect for medicinal recovery
So during this unfortunate pandemic of the Covid-19 crisis, I brought an industrial UV light in to our lab at work, one that kills bacteria, germs and viruses in less than a minute. ****** me got exposed for about 3+ minutes at close proximity, not thinking nor ever experienced the consequences of being exposed from one of these devices before. Couple of hours later I started to develop sunburn on one side of my body but even worse my eyes turned red like a rabbit and it felt like bumps all over my eyes. Later that evening my eyes were in severe pain and I could not even open them. My eyes were literally sunburned! Because of the global pandemic, I'm in a city where an emergency Dr. visit would definitely not be a priority nor really welcomed. I remembered I had some actovegin stored away and decided to take one. Immediately I started having energy and my eyes started feeling better as well. 3 to 4 hours later I took another one, I regained my appetite and could open my eyes again. 3 to 4 hours after the second dose, I took another one and was amazed at the rapid recovery I was making. A little over 24hrs after my idiotic mistake, I gained a lesson learned and felt almost back to baseline normal! I'm really amazed at how fast I went from one extreme to another in such a short period. I read sources that said expect discomfort for several days or even weeks from intense UV exposure. I've taken actovegin before but never really had a genuine need for it. I've used it a couple times for extra endurance during hikes and exercise, but I now have a new respect for it and I'm glad I bought a few bottles being readily available for when I REALLY needed it than not having any and still in extreme pain wishing I had. Recovery like this is a true testament to the medicinal value of this product! Thanks cosmicnootropic for making it available, reasonable price and authentic!
01/23/2020
Savannah B.
Verified BuyerActovegin
Original order did not make it to US, but cosmicnootropic quickly reached out to ask whether I wanted a refund or secondary shipment. New shipment arrived promptly.
09/28/2019
Gerrard H.
Verified BuyerHealing
It’s amazing for site injection injury’s heals so much faster
07/29/2019
Inna T.
Verified BuyerActovegin perfect quality
Super! Fast shipping. Good product. Will buy more stuff in fall when it is not so hot outside so the medicine will not spoil in the heat.
03/25/2019
Kurtis M.
Verified BuyerExcellent
I've had two knee surgeries and Actovegin has really helped me get back in the gym.
11/08/2018
brian i.
Verified BuyerExcellent as always
Love dealing with these people. Always helpful.
06/16/2018
Adrian
Verified BuyerOne of the best PED and the most underrated
I used both injections and pills. For the pills, I took 3 pills 3 times a day. For me it works wonder, it gives me better cardio, better recovery and less muscle pain. I think this product is awesome ! The injection are better than the pills but you have to use at least 5ml 3 times a week which is a lot. You can use it IM or IV. Pill take a little more time to work than injection and cost more but you will feel the effects for sure. Enjoy !
01/01/1970
Thank you Adrian! Hopefully this feedback will help others with choosing right variant of Actovegin
05/27/2018
brian i.
Verified BuyerThis company and this product is awesome!
Both tte compnany and the Actovegin exeeded my expectations. Looking forward to deaingl with Cosmic Nootropic more in the future.
Thank you!
You will now receive regular updates from us!
Your coupon