Card payments are temporarily suspended for technical reasons! Please consider paying with cryptocurrencies with 15% off.
Card payments are temporarily suspended for technical reasons! Please consider paying with cryptocurrencies with 15% off.
days left












AUTHENTIC PRODUCTS
Only original pharmaceutical products that passed 3 stages of clinical trials.

GMP QUALITY
Our products are produced on GMP or state licensed factories.

10% CRYPTO DISCOUNT
10% discount for paying in BTC or other cryptocurrency.

PAYMENT BY CARD
Convenient Payment by Visa or MasterCard.

GUARANTEED DELIVERY
Guaranteed delivery times and clear Reshipment & Refund Rules.

PROMPT SHIPPING
Different shipping methods and same or next day shipping.
CEREBROLYSIN ®
277 Reviews
Buy Cerebrolysin Online!
Cerebrolysin is a mix of enzymatically treated neuropeptides (20%) and amino acids (80%) derived from pig brains. Its constituents can include low molecular weight neuropeptides that are similar to fragments of natural neurotrophic factors (NTF), for example, ciliary neurotrophic factor (CNTF), brain-derived neurotrophic factor (BDNF), glial cells derived neurotrophic factor (GDNF), nerve growth factor (NGF), and insulin-like growth factor (IGF).
Cerebrolysin has been studied for potential use in a wide variety of neurodegenerative disorders, such as dementia including Alzheimer’s disease, stroke, and traumatic brain injury (TBI).
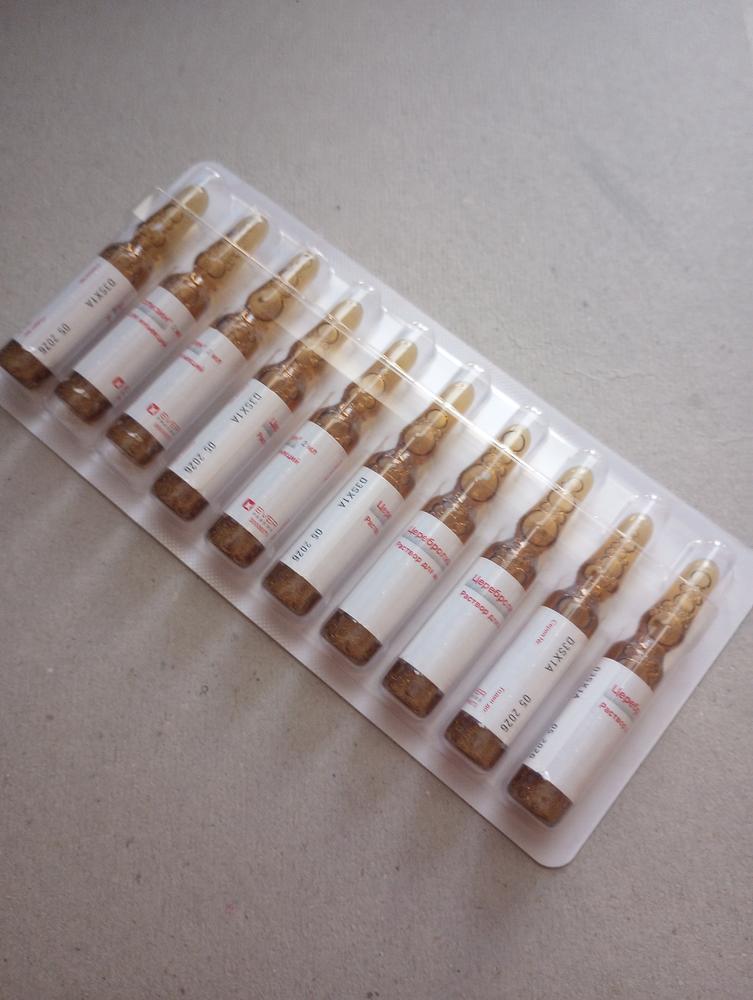
Cerebrolysin is available in different ampoule sizes: from 2ml to 20ml. We offer worldwide shipping!
| Generic name: | Cerebrolysin, FPF-1070 |
| Form: | Solution for intramuscular and intravenous injections in ampoules/vials. |
| Form description: | Transparent solution of amber color |
| Active ingredient: | 215.2 mg of Cerebrolysin concentrate in an aqueous solution |
| Excipients: | Sodium hydroxide and water for injection |
| СAS Number: | 12656-61-0 |
| ATC code: | N06ВХ Nootropics |
| Shelf life: | 5 years. Do not use it beyond the expiration date printed on the package. |
| Storage: | Store at temperatures not exceeding 25°C (77°F). Do not freeze. Protect from light. |
| Manufacturer: | EVER Pharma, Austria. www.everpharma.com |
What is Cerebrolysin Used For?
Cerebrolysin is a neuropeptide preparation with neuroprotective and neurorestorative effects. In some countries this “smart drug” is used in the complex treatment of cerebrovascular disorders:
- Strokes and stroke complications;
- Alzheimer’s disease and other types of dementia;
- Traumatic brain injuries;
- Spinal cord injuries;
- ADHD in children;
- Antidepressant-resistant depressions;
- Other brain diseases.
What are Cerebrolysin Benefits?
According to the manufacturer, Cerebrolysin is used in the complex treatment of acute and chronic disorders of the central nervous system (CNS). It supports endogenous repair and recovery processes after an injury or degenerative disease. It also helps to protect the brain against pathological events and cascades. Cerebrolysin mechanism of action resides in its ability to activate endogenous defense mechanisms (ESM), which include four main components: 1) Neurotrophic, 2) Neuroprotection, 3) Neuroplasticity, and 4) Neurogenesis.
As practice shows, repeated and long-term courses of treatment with Cerebrolysin peptide lead to an increase in the effectiveness of neuro-therapy, especially in relation to improved cognitive and behavioral functions.
In clinical practice, Cerebrolysin is known to reduce symptoms of depression, cognitive impairment, and autonomic disorders. It is said to relieve pain syndrome and enhance the effect of tricyclic antidepressants.
Is Cerebrolysin Safe to Take?
Cerebrolysin has an excellent safety record. Unless taken in large amounts (>10 ml), adverse reactions are mild and rare.
Special attention shall be taken when stacking Cerebrolysin with antidepressants, including MAO inhibitors.
Where To Buy Cerebrolysin?
Authentic Cerebrolysin ampoules for injection are available at CosmicNootropic. We offer the most popular forms: Cerebrolysin 5ml and Cerebrolysin 10ml ampoules as well as other volumes depending on the purpose of use.
You do not need a prescription to obtain Cerebrolysin online. Buy it with worldwide delivery. CosmicNootropic offers bulk discounts 👍
Check out our blog post
🧠 Comprehensive Cerebrolysin Review with FAQ Section 🧠
Legal Disclaimer
This product has not been approved by the US FDA. All statements on this page are for informational purposes only and have not been evaluated by the US FDA.
This product is not intended to diagnose, treat, cure, or prevent any disease. See more
What is the Optimal Cerebrolysin Dosage?
Cerebrolysin has indication-specific dosing. The most common regime for improving brain function is IM injections of 5 ml daily. The course of treatment is 4 weeks unless otherwise prescribed by your attending physician.
Dose recommendations from the manufacturer:
| Condition | Dosage | When to start | Duration |
| Stroke | 20 – 50 ml | / as soon as possible | / 10 – 21 days |
| Vascular dementia | 10 – 30 ml | / as soon as possible | / 1 cycle: 5 days a week/ 4 weeks, 2-4 cycles per year |
| Alzheimer’s disease | 10 – 30 ml | / as soon as possible | / 1 cycle: 5 days a week/ 4 weeks, 2-4 cycles per year |
| TBI | 20 – 50 ml | / as soon as possible | / 7 – 30 days |
How to Inject Cerebrolysin?
The main ways of administering the drug are intramuscular (IM) and intravenous (IV). The gluteal region is the best spot to inject IM. Relax your muscle and inject slowly. Here are a couple of tips on how to make injections. Please follow the sterility instructions!
What are Cerebrolysin Side Effects?
Rare cases of adverse reactions to Cerebrolysin may include fatigue, headaches, dizziness, insomnia, etc.
Please note that it is important to follow the recommendations of the drug administration to minimize the chance of adverse reactions.
The drug is injected slowly. If the injection is made too fast, in rare cases, there can be a feeling of heat, sweating, dizziness, heart palpitations, or arrhythmia.
Cerebrolysin is reported to lose its effectiveness when used for longer than necessary. However, there are anecdotal cases of Cerebrolysin causing anxiety, headaches, and aches in the body when the medication brings no benefit any longer.
CEREBROLYSIN OFFICIAL INSTRUCTION
- Rutheret al (1994) Efficacy of the Peptidergic Nootropic Drug Cerebrolysin in Patients with Senile Dementia of the Alzheimer Type (SDAT) https://www.ncbi.nlm.nih.gov/pubmed/8159781
- L. Francis-Turner, V. Valouskova (1996) Nerve growth factor and nootropic drug Cerebrolysin but not fibroblast growth factor can reduce spatial memory impairment elicited by fimbria-fornix transection: short-term study https://www.ncbi.nlm.nih.gov/pubmed/8848264
- Schwab et al (1997) Morphofunctional effects of moderate forebrain ischemia combined with short-term hypoxia in rats – protective effects of Cerebrolysin https://www.ncbi.nlm.nih.gov/pubmed/9085071
- V. Valouskova and A. Gschanes (1999) Effects of NGF, b-FGF, and Cerebrolysin on Water Maze Performance and on Motor Activity of Rats: Short- and Long-Term Study https://www.ncbi.nlm.nih.gov/pubmed/10082636
- Masliah et al (1999) Cerebrolysin Ameliorates Performance Deficits, and Neuronal Damage in Apolipoprotein E-Deficient Mice https://www.ncbi.nlm.nih.gov/pubmed/9972690
- Bae et al (2000) A Double-Blind, Placebo-Controlled, Multicenter Study of Cerebrolysin for Alzheimer’s Disease https://www.ncbi.nlm.nih.gov/pubmed/11129744
- Ruther et al (2000) Sustained improvements in patients with dementia of Alzheimer’s type (DAT) 6 months after termination of Cerebrolysin therapy https://www.ncbi.nlm.nih.gov/pubmed/11005546
- Ruether et al (2001) A 28-week, double-blind, placebo-controlled study with Cerebrolysin in patients with mild to moderate Alzheimer’s disease https://www.ncbi.nlm.nih.gov/pubmed/11552768
- Hartbauer et al (2001) Antiapoptotic effects of the peptidergic drug Cerebrolysin on primary cultures of embryonic chick cortical neurons https://www.ncbi.nlm.nih.gov/pubmed/11475013
- Hartbauer et al (2001) Effects of Cerebrolysin on the outgrowth and protection of processes of cultured brain neurons https://www.ncbi.nlm.nih.gov/pubmed/11459078
- Eder et al (2001) Increased density of glutamate receptor subunit 1 due to Cerebrolysin treatment: An immunohistochemical study on aged rats https://www.ncbi.nlm.nih.gov/pubmed/12197668
- Panisset et al (2002) Cerebrolysin in Alzheimer’s disease: a randomized, double-blind, placebo-controlled trial with a neurotrophic agent https://www.ncbi.nlm.nih.gov/pubmed/12111446
- Rockenstein et al (2003) The neuroprotective effects of Cerebrolysin in a transgenic model of Alzheimer’s disease are associated with improved behavioral performance https://www.ncbi.nlm.nih.gov/pubmed/14628195
- Ladurner et al (2004) Neuroprotective treatment with Cerebrolysin in patients with acute stroke: a randomised controlled trial https://www.ncbi.nlm.nih.gov/pubmed/15583955
- Alvarez et al (2006) A 24-week, double-blind, placebo-controlled study of three dosages of Cerebrolysin in patients with mild to moderate Alzheimer’s disease https://www.ncbi.nlm.nih.gov/pubmed/16420392
- Rockenstein et al (2006) Cerebrolysin Decreases Amyloid-b Production by Regulating Amyloid Protein Precursor Maturation in a Transgenic Model of Alzheimer’s Disease https://www.ncbi.nlm.nih.gov/pubmed/16511867
- Wei et al (2007) Meta-analysis: the efficacy of nootropic agent Cerebrolysin in the treatment of Alzheimer’s disease https://www.ncbi.nlm.nih.gov/pubmed/17318304
- Ren et al (2007) Cerebrolysin enhances functional recovery following focal cerebral infarction in rats https://www.ncbi.nlm.nih.gov/pubmed/17473393
- Alvarez et al (2008) Reductions in qEEG slowing over 1 year and after treatment with Cerebrolysin in patients with moderate–severe traumatic brain injury https://www.ncbi.nlm.nih.gov/pubmed/18273537
- Muresanu et al (2008) A pilot study to evaluate the effects of Cerebrolysin on cognition and qEEG in vascular dementia: Cognitive improvement correlates with qEEG acceleration https://www.ncbi.nlm.nih.gov/pubmed/18048059
- Alvarez et al (2009) Reduced TNF-a and increased IGF-I levels in the serum of Alzheimer’s disease patients treated with the neurotrophic agent Cerebrolysin https://www.ncbi.nlm.nih.gov/pubmed/19531281
- Sharma et al (2010) Cerebrolysin reduces blood-cerebrospinal fluid barrier permeability change, brain pathology, and functional deficits following traumatic brain injury in the rat https://www.ncbi.nlm.nih.gov/pubmed/20633118
- Guekht et al (2011) Cerebrolysin in Vascular Dementia: Improvement of Clinical Outcome in a Randomized, Double-Blind, Placebo-Controlled Multicenter Trial https://www.ncbi.nlm.nih.gov/pubmed/20656516
- Alcantara-Gonzalez et al (2012) Combined Administration of Cerebrolysin and Donepezil Induces Plastic Changes in Prefrontal Cortex in Aged Mice https://www.ncbi.nlm.nih.gov/pubmed/22826038
- Heiss (2012) Cerebrolysin in Patients With Acute Ischemic Stroke in Asia Results of a Double-Blind, Placebo-Controlled Randomized Trial https://www.ncbi.nlm.nih.gov/pubmed/22282884
- Menon et al (2012) Cerebrolysin, a Mixture of Neurotrophic Factors Induces Marked Neuroprotection in Spinal Cord Injury Following Intoxication of Engineered Nanoparticles from Metals https://www.ncbi.nlm.nih.gov/pubmed/22229324
- Chen et al (2013) Cerebrolysin enhances cognitive recovery of mild traumatic brain injury patients: double-blind, placebo-controlled, randomized study https://www.ncbi.nlm.nih.gov/pubmed/23656173
- Amiri-Nikpour et al (2014) Cerebrolysin effects on neurological outcomes and cerebral blood flow in acute ischemic stroke https://www.ncbi.nlm.nih.gov/pubmed/25516711
- Muresanu et al (2015) Cerebrolysin and Recovery After Stroke (CARS) A Randomized, Placebo-Controlled, Double-Blind, Multicenter Trial https://www.ncbi.nlm.nih.gov/pubmed/26564102
- Ziganshina et al (2016) Cerebrolysin for acute ischaemic stroke https://www.ncbi.nlm.nih.gov/pubmed/27918088
- Chutko et al (2017) [The efficacy of cerebrolysin in the treatment of autism spectrum disorders] https://pubmed.ncbi.nlm.nih.gov/29053124/
- M Brainin (2018) Cerebrolysin: a multi-target drug for recovery after stroke https://pubmed.ncbi.nlm.nih.gov/30004268/
- Torshin et al (2019) [Cerebrolysin peptides as mood stabilizers] https://pubmed.ncbi.nlm.nih.gov/31994517/
- Alzoubi et al (2020) Cerebrolysin prevents sleep deprivation induced memory impairment and oxidative stress https://pubmed.ncbi.nlm.nih.gov/31987894/
- S Gavrilova, A Alvarez (2021) Cerebrolysin in the therapy of mild cognitive impairment and dementia due to Alzheimer’s disease: 30 years of clinical use https://pubmed.ncbi.nlm.nih.gov/32808294/
- Strilciuc et al (2021) Safety of Cerebrolysin for Neurorecovery after Acute Ischemic Stroke: A Systematic Review and Meta-Analysis of Twelve Randomized-Controlled Trials https://pubmed.ncbi.nlm.nih.gov/34959697/
- Mureșanu et al (2022) The Effect of Cerebrolysin on Anxiety, Depression, and Cognition in Moderate and Severe Traumatic Brain Injury Patients: A CAPTAIN II Retrospective Trial Analysis https://pubmed.ncbi.nlm.nih.gov/35630065/
- Villarreal-Reyna et al (2022) Cerebrolysin induces hair repigmentation associated to MART-1/Melan-A reactivation https://pubmed.ncbi.nlm.nih.gov/36411485/
4.6
Based on 277 Reviews
(234)
(12)
(4)
(6)
(21)

04/24/2024
Johan S.
Verified Buyer
NO
Never got it
Because Cosmic write whats actually in the package on the package, my country wouldn't allow these items through customs. Just write that they are supplements and it'd be fine...
04/26/2024
Hi Johan! Thank you for leaving your review. We are so sorry to hear that you never received your order. It looks like we tried to do everything we can to help your parcel get through the customs. We have emailed you to see if there's anything else we can do in this situation, please check your inbox.
04/04/2024
David V.
Verified Buyer
US
I didn’t receive it
Even though the post record that it been delivered we looked everywhere and we didn’t find it
04/04/2024
Dear David, we are so sorry to hear this. We have contacted you via email to see what can be done in this situation.
04/01/2024
Deborah W.
Verified Buyer
US
Cerebrolysin
Fabulous! I can feel the positive benefits in a short time. Helping brain issues I’ve had with memory + depression.
04/02/2024
Hi Deborah! We are so happy to hear that Cerebrolysin has had a positive impact on your life. Thank you for sharing!
03/30/2024
Dustin S.
Verified Buyer
US
Cerebrolysin
Great! Happy with the cerebrolysin. No bad reactions. Starting off at only 2ml a day before bed. Will order the 5 ml next time.
03/31/2024
Thanks for your review, Dustin! We're glad to hear that you're happy with Cerebrolysin and that you haven't experienced any side effects. And starting with 2ml a day at bedtime sounds like a sensible approach 🙌️
03/14/2024
Chadi A.
Verified Buyer
DK
Fast delevery
I was so Happy for The fast delevery, and The produkt as Well, thank you a lot, best experience with you.
03/15/2024
Dear Chadi, thank you so much for your kind feedback!
03/04/2024
Olga M.
Verified Buyer
US
Love the product Received what I needed and on time Thank you
03/07/2024
Dear Olga, thank you for your feedback! Glad to see you as our customer again :)
03/03/2024
ERIC M.
Verified Buyer
US
Excellent product!
Wasn't sure how the Cerebralysom would work, but I have now bought multiple times and will purchase more!
03/07/2024
Dear Eric, your positive experience and preference mean the world to us. Thank you for your feedback!
03/02/2024
Edward M.
Verified Buyer
US
Greatest peptide of all time
I stopped biting my fingernails and all nervous behaviors without even trying. It killed my anxiety
03/03/2024
Dear Edward, What fantastic news! We're happy to hear that Cerebrolysin has not only helped you stop nervous behaviors but also alleviated your anxiety. Thank you for choosing Cerebrolysin and for sharing your review with us!
03/02/2024
Jasmin B.
Verified Buyer
US
Amazing
Loved the product and I noticed a huge difference with my memory. It’s like my brain worked again!! Lol
03/03/2024
Dear Jasmin, We're thrilled to hear about the positive impact on your memory! It's fantastic that you've experienced such noticeable results.
03/01/2024
MICHAEL U.
Verified Buyer
US
It works
Probably the #1 thing that's helped my depression and anxiety.
03/01/2024
Dear Michael, we are beyond happy to hear that this product has helped you!
02/23/2024
Violet E.
Verified Buyer
US
CEREBROLYSIN or Noben:or Was it Both?
Hi. I tried both CEREBROLYSIN then Noben right after. Dementia runs on both sides of my family. I noticed I was super forgetful and it was difficult to form coherent sentences. I work with customers and manage a team so my communication needs to be on point. So I decided to try some things to help. I'm not sure if it was CEREBROLYSIN or Noben that helped lift the fog away. Now it's a natural flow to talk with little effort to think about what to say or respond. I feel my confidence back and my stress levels have dropped. I have my life back! Thank you!
02/25/2024
Dear Violet, Thanks for sharing your experience with these products! It's great to hear that they've helped improve your cognitive function and overall well-being. We're thrilled to have contributed to bringing back confidence and reducing stress in your life. Wishing you continued success!
02/10/2024
Theodore H.
Verified Buyer
US
Excellent service!
Top notch. Will buy again!
02/15/2024
Thank you, Theodore! Looking forward to see you as our customer again soon!
02/02/2024
scott c.
Verified Buyer
US
Cerebrolysin was Great!
They had some problems with United States Shipping. Had to wait 3 weeks...however my mind is MUCH brighter and I am much more positive. 20ML made me a believer. Cosmic will get my business again.
02/02/2024
Hi Scott! We are sorry that the shipping was not as smooth. We hope it will not happen again. Thank you for leaving your review and sharing your experience with Cerebrolysin with us!
01/31/2024
Lyn T.
Verified Buyer
AU
Cerebrolysin
Noticeable difference in finding my words as a 72 yr old
02/01/2024
Dear Lyn, we are happy that our product has had a positive impact on your life!
01/31/2024
Rhonda B.
Verified Buyer
US
Cerebrolysin
There is no better source. Thank you!
02/01/2024
Hi Rhonda! Thank you so much for your kind words!
01/22/2024
Stephen C.
Verified Buyer
US
Great company
You guys are amazing. Quick shipping!!!
01/26/2024
Dear Stephen, thank you for your kind words! We really appreciate it! 🤗
01/22/2024
Richard P.
Verified Buyer
US
Works great just realize it bites back
Product works great however, I found it really smarts on administration. Benefits out way the momentary flopping on the floor, grabbing your buttocks or crying like a newborn. Seriously, its a great product and highly recommend.
01/26/2024
Dear Richard, thank you for the review! We are glad that you are satisfied with our product :)
01/12/2024
Christina E.
Verified Buyer
US
Not sure
I haven’t noticed a real difference yet, but I’m hopeful! Great delivery & pricing.
02/07/2024
Dear Christina, thank you for the feedback! We're glad you were satisfied with our service. Please note that the effect of each product may vary between individuals.
01/10/2024
Siddharth S.
Verified Buyer
TH
Great
Quickly posted and received in 3 weeks!
01/12/2024
Hi Siddharth! We are happy that you are satisfied with our service! :)
01/09/2024
Chad T.
Verified Buyer
US
Been a Week Since Ordering.. No word of anything
Hi, Let me just start off by stating that I am a huge fan of Cosmic Nootropic. I love the Cerebrolysin product, it works extremely well and I am happy to be a proud customer of Cosmic Nootropic (I recommend it to my friends for Christ' sake..). That was until now. I ordered Cerebrolysin a week ago and haven't seen it move at all with the shipping, which is very upsetting. I pay good money for the cerebrolysin, which I need very urgently for privatized reasons, and I have not heard anything from the Cosmic Nootropic team nor have I seen a status update on the USPS website. It is just disgruntling because I am very good for my end of the deal-- even flourishing Cosmic Nootropic with new clientele and yet I can't even get a "hey, we are sorry for the delay, here is where your package is". Oh well. Hopefully Cosmic Nootropic can redeem themselves and regain my trust.
01/12/2024
Dear Chad, We're sorry to hear that you still haven't received your order. Since the order has already been forwarded to USPS, we cannot influence the speed of its delivery. We have contacted you via email to resolve this unpleasant situation, please check!
Thank you!
You will now receive regular updates from us!
Your coupon