Piracetam Review 2023: Best Memory Supplement, Different Racetams, Benefits & Side effects of Piracetam (Nootropil)
September 3, 2023
PIRACETAM REVIEW TABLE OF CONTENTS:
- What is Piracetam?
- Piracetam pharmacodynamics
- What are Piracetam benefits and effects?
- Piracetam side effects
- Piracetam intended uses
- Piracetam research
- Piracetam stacks
- How to take Piracetam?
- Piracetam course: how long to take?
- Piracetam injections or pills?
- Can you drink alcohol while taking Piracetam?
- How to choose the right Piracetam dosage?
- What are the best racetams?
- Piracetam sources
- Piracetam review on Reddit
- Conclusion
- Bibliography
WHAT IS PIRACETAM?
Piracetam (or Nootropil) is the touchstone of all nootropic drugs and an ancestor of the nootropic racetam family. It was synthesized in 1964 and still remains one of the most important chemical compounds in the group.
It is frequently prescribed to older patients, children, and adults for brain enhancement and relief of the following symptoms:
- Memory disorders,
- Psychoorganic syndrome with asthenia, impaired attention, speech,
- Headaches and dizziness,
- Alcoholic psychoorganic syndrome,
- Treatment-resistant depressions,
- Other conditions characterized by impaired mnestic function including Alzheimer’s disease.
Also Piracetam is often used by healthy individuals searching for safe and mild cognitive improvement.
PIRACETAM PHARMACODYNAMICS
Chemically, Piracetam is a derivative of pyrrolidone. Its chemical structure is similar to the GABA neurotransmitter and it is sometimes considered a derivative of this amino acid (Pic. 1). Nevertheless Piracetam is not transformed into GABA in the body and the GABA content in the brain is not increased after using Piracetam. Still in relatively high doses and when administered repeatedly it can enhance GABA-ergic inhibition processes [43, 32].
Picture 1: 3D model and chemical formula of Piracetam
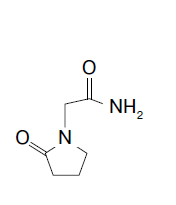
Piracetam has an important effect on neurotransmission, which is not limited to any single type of neurotransmitter. The drug interacts with a system of neurotransmitters, modulating the cholinergic [46, 11, 18, 4], serotonergic [3], noradrenergic [5] and glutamatergic [29, 39] neurotransmission, which is especially important since impaired synaptic transmission involving acetylcholine and glutamate is responsible for age-related disorders of memory and other cognitive functions [46, 29, 36].
The drug has multidirectional effects on the main neurotransmitter systems in different parts of the brain. For example, Piracetam significantly increases the serotonin (5-NT) content in the frontal cortex and decreases it in the striatum, hypothalamus and brainstem [3]. Dopamine level under Piracetam is increased in the frontal cortex and striatum [5]. The effect of Piracetam on the cholinergic system of the brain is manifested by an increase in the synthesis and release of acetylcholine, choline reuptake in the brain, and an increase in the sensitivity and number of muscarinic receptors [30].
The main mechanism of action of Piracetam, as well as many other nootropics, is associated with changes in metabolic, bioenergetic processes in the nerve cell, an increase in the rate of turnover of informational macromolecules and activation of protein synthesis [13, 46]. As a result, Piracetam exhibits a pronounced neuroprotective and anticonvulsant effect and improves neuroplasticity. At the vascular level, Piracetam arguably reduces platelet aggregation by increasing erythrocyte deformability. It decreases erythrocyte adhesion to vascular endothelium, viscosity of plasma and whole blood, inhibits the development of vascular spasms and facilitates microcirculation [28, 31, 34, 33, 46, 1, 32].
WHAT ARE PIRACETAM BENEFITS AND EFFECTS?
Studies in experimental pharmacology have highlighted the main pharmacological effects inherent in Piracetam. Arguably it can:
- Improve blood circulation in ischemic areas, and metabolism in the brain;
- Increase the resistance of the brain tissue to hypoxia and toxic effects;
- Enhance potential neurophysiological capabilities;
- Activate mental activity (brainwork, learning, memory, etc.);
- Restore and stabilize brain functions;
- Improve mood, attention and memory;
- Exhibit a certain anticonvulsant effect;
- Correct CNS dysfunctions and neurological deficits resulting from aging.
Piracetam is often used by healthy individuals searching for safe and mild cognitive improvement. But Piracetam is mostly prescribed to older patients in order to maintain mental abilities.
PIRACETAM SIDE EFFECTS
Piracetam is a safe, low toxic nootropic drug. Almost all studies report high tolerability to the compound.
Still there can be some side effects to it. They may include such adverse reactions as sleep disturbances, dyspeptic disorders, agitation, as well as sexual stimulation and increased motor activity. That is why it is not recommended to take Piracetam in the evening, before bed-time. In addition it is advised against driving vehicles and operating machinery while on Piracetam because of the above-mentioned possible adverse reactions.
Headache is another adverse reaction that may occur while taking Piracetam. It can be explained by choline deficiency intrinsic in our bodies. If this is the case, it is advised to add Citicoline to the stack.
Consult your doctor before taking Piracetam if you are pregnant, breastfeeding, have renal impairment or any other medical conditions. More information is available in the official Piracetam instruction on the product page.
PIRACETAM INTENDED USES
The main indications for the use of Piracetam include [46]:
- Cerebral circulation disorders (intensive therapy of ischemic stroke and rehabilitation after it, decompensation of chronic insufficiency of cerebral circulation, acute phase of traumatic brain lesions, comatose states);
- Disorders of memory and other higher cognitive functions (aging, dementia, mental retardation, learning difficulties);
- Speech disorders (aphasia, dyslexia (special difficulties in learning written speech) in children);
- Dizziness;
- Encephalopathies (alcohol and opium addiction, drug intoxication, consequences of neuroinfections, post-hypoxic conditions);
- Psychosis, astheno-depressive states, apathetic states in schizophrenia, depressive states resistant to antidepressants (in complex therapy), correction of side effects and complications of therapy with psychotropic agents.
It is necessary to note that the treatment of the above mentioned conditions shall be complex and conducted under the doctor’s supervision.
PIRACETAM RESEARCH
Over the last decades, Piracetam has been extensively studied and it is currently approved in over 100 countries. In particular major scientific attention has been given to the research of the treatment of age-related disorders with Nootropil (Piracetam).
Thus, in a prospective randomized placebo-controlled double-blind study [9] 130 patients suffering from psychoorganic syndrome in dementia were treated with Piracetam at a dose of 4800 mg/day for 3 months.
The study methods included: the Clinical Global Impression Scale (CGI) filled out by physicians, the Sandoz Clinical Assessment Geriatric Scale (SCAG) used by clinical psychologists, and the BGP Geriatric Assessment Scale filled out by the nursing staff. In addition, the short syndrome STK test and the Benton test were used. The results of the study showed an improvement of 50% or higher (p<0.001) in three out of four examined indicators, whereas in placebo groups these values were 0-6%.
Another 6-month double-blind placebo-controlled study [15] enrolled 81 patients with common symptoms of moderate senile Alzheimer-type dementia. The first group received Piracetam at 6 g per day for the first month and 3 g per day for the next 5 months. The second group received placebo during the whole period.
Geriatric assessment scales and psychometric tests were used to evaluate the effectiveness of the treatment. The results of the study indicated that Piracetam significantly increased the initial scores in the psychometric tests compared to placebo. A particularly significant difference between Piracetam and placebo was evident in the behavioral tests, allowing the authors to conclude that Piracetam can improve patient self-reliance and communication with the world.
Of particular interest are large multicenter studies of the effectiveness of Piracetam in the elderly patients with a variety of neuropsychiatric symptoms. It should be noted that the use of Piracetam, along with improvements in the cognitive sphere, was accompanied by a reduction in the concomitant psychopathological symptomatology – depressive and other disorders.
In an open multicenter study of 5306 patients aged 14-96 years old [46] with symptoms of cerebrovascular insufficiency took Piracetam at 1200 mg/day for 4 weeks. After 4 weeks of therapy a general improvement of the condition was observed in 88% of patients, no changes – in 11.5%, deterioration – in 0.5%. Concomitantly:
- the number of patients with vertigo decreased from 3265 to 960 (an improvement of 70.6%),
- with decreased motivation – from 3803 to 1185 (68.8%),
- with pronounced fatigue – from 4255 to 1474 (65.4%),
- with depression – from 2238 to 830 (62.9%),
- with decreased concentration – from 4676 to 1759 (62.4%),
- with decreased adaptation – from 1566 to 623 (60.2%),
- with tinnitus – from 2064 to 888 (57%),
- with impaired perception – from 4518 to 2086 (53.8%),
- with sleep disturbance – from 3024 to 1690 (44.1%).
Tolerability of the drug was rated as very good: only 339 (6.6%) patients had minor side effects – sleep disturbances, gastrointestinal disturbances, agitation, headaches.
Similar data were obtained in another multicenter study that evaluated the efficacy of Piracetam in 11654 patients. Their average age was 64 years old. [46] More than 70% of patients had memory impairments. Other disorders included dizziness (650 patients), arterial hypertension (423), headaches (320) and depression (213). Piracetam was administered for 90 days in different dosages: in patients younger than 60 years old – 2 capsules 3 times daily (2400 mg/day) and in patients older than 60 – a drinking solution of 1 g three times daily (3 g/day). The patients were examined before the start of the therapy, on the 45th and 90th day of treatment. The results of the study showed high effectiveness of Piracetam: 70% of doctors and 90% of patients noted positive effects of the drug and only 7% of patients complained about side effects.
Piracetam has been successfully used in the treatment of chronic alcoholism (withdrawal states) and alcoholic delirium. Fischer et al [46] studied 97 patients with acute alcohol withdrawal. All of them received Piracetam by infusion in a dose of 12 g/day for 3-5 days in hospital conditions. Researchers assessed initial severity of such symptoms as tremor, anxiety, hyperhidrosis, nausea, anxiety and depression after 30 min, and 1, 3 and 5 days after the start of the therapy with Piracetam.
The results of the study showed quite high effectiveness of Piracetam in the study group – “very good” in 57%, “good” in 30%, “satisfactory” in 11% and “unsatisfactory” in only 2% of the cases. Significant improvement in all symptoms was noted as early as 30 min after the beginning of the treatment with Piracetam.
This study allowed the authors to conclude that it is advisable to use Piracetam in the therapy of acute alcohol withdrawal due to the rapid onset of a positive effect, good tolerance and the possibility of reducing benzodiazepines doses. In case of ineffectiveness of such treatment the authors recommended combining it with tiapride, tranquilizers or anticonvulsants.
PIRACETAM STACKS
Many studies have been devoted to combining Piracetam with other psychotropic medications.
The effectiveness of the combined use of Piracetam and anticonvulsants (carbamazepine, clonazepam, valproate) has been proven in myoclonus of various genesis and myoclonus-epilepsy. Despite the fact that Piracetam has no anticonvulsant effect of its own, most researchers note the ability of the drug to reduce the number of epileptic seizures and reduce myoclonus manifestations, and to have a positive effect on neurological symptoms particularly ataxia, motor, writing and eating disorders. In addition, against the background of using Piracetam patients’ motivation and attention increased, and depression and insomnia manifestations lowered. [46]
Regarding the vaso-autonomic part of the psychopharmacological action of the drug, its effect on autonomic symptoms in 50 patients with various forms of resistant depression was investigated. Piracetam in combination with antidepressants (Amitriptyline and Lyudiomil) was administered at an average daily dose of 1600 mg. The findings were compared with those of the control group who received antidepressants only. The main group showed greater reduction of vegetative symptoms compared to the control group, especially patients with emotional and motor retardation. [46]
Positive data were obtained when using Piracetam with neuroleptics (protective effect) as an agent that can prevent the development of extrapyramidal disorders. While Piracetam like other GABAergic agents, has no antiparkinsonian effect, due to its GABAergic action it can play a certain role in relieving extrapyramidal disorders. So, Piracetam was prescribed with neuroleptics and correctors in 28 patients with subacute extrapyramidal syndrome. The initial dose of 1.2 g was gradually increased until a clear therapeutic effect was achieved. Usually a noticeable improvement occurred on the 5th-6th day of therapy at the dose of 3.2-4 g, and by the 7th-10th day the extrapyramidal syndrome was completely eliminated. [46]
According to the official Piracetam instructions the possibility of changing the pharmacodynamics of the compound under the influence of other drugs is low, because 90% of its dose is excreted in the urine unchanged.
HOW TO TAKE PIRACETAM?
The standard Piracetam dosage is 800 mg / three times a day, which is 2.4 g in total. For cognition-related issues, it can be increased up to 4.8 g/daily. The exact dosing of Piracetam depends on the person’s condition and the desired effects.
It is not recommended to take Piracetam at night because of a possible stimulating effect. Doctors advise taking one dose in the morning, another in the afternoon, and the last one after lunch before 5 pm. To read more on Piracetam doses please refer to the official instructions.
PIRACETAM COURSE: HOW LONG TO TAKE?
One pack of Piracetam contains 30 pills, each with 800 mg of the active ingredient. It is sufficient for an average of 10 days of supplementation.
If Piracetam is used as a mild cognitive enhancer it shall be normally taken as a course from 4 to 6 weeks. In certain cases it can be taken as long as 3-4 months. The main effects usually begin to manifest after 3-4 weeks of supplementation.
PIRACETAM INJECTIONS OR PILLS?
Piracetam is available as an injection and in a pill form. Which one is better? It highly depends on the disorder. In acute conditions, doctors prescribe Piracetam injections. Piracetam capsules are indicated for a longer course or for rehabilitation therapy.
Piracetam injection dosage shall be determined individually according to the instructions and doctor’s recommendations.
CAN YOU DRINK ALCOHOL WHILE TAKING PIRACETAM?
Doctors do not recommend combining Piracetam and alcohol, because such a combination may overstimulate the nervous system. It is advised to refrain from alcohol while treating the nervous system.
HOW TO CHOOSE THE RIGHT PIRACETAM DOSAGE?
It is interesting to study how effective Piracetam is depending on the dosage. In fact the range of recommended doses of Piracetam varies widely: from 1 to 45 g per day. And good tolerability of the drug makes it possible to quickly increase the dose. However, the average dosage range of Piracetam usually goes from 1.2 to 9.6 g/day.
According to one concept, every nootropic drug has a limited “therapeutic window” in which medium doses are the most effective, while low and high doses turn out ineffective. This perspective was confirmed in a study of healthy volunteer operators who took different doses of Piracetam: single intake of 2.9, 4.8 or 9.6 g/day. The results demonstrated reliably higher performance indicators and fewer errors in operators who took 4.8 g of Piracetam.
In another method of choosing how much Piracetam to take, the focus was given to the total course dosage rather than the daily one. When studying the efficacy of Piracetam in 138 patients with various mental disorders (schizophrenia, bipolar, depression, TBI, atherosclerotic psychoses, alcoholism, neurosis) it was found that the positive therapeutic effect was achieved with the course dose of 50-64 g, which is about 62-80 pills of Piracetam 800mg. This approach to determine the effective dose of the drug allows reducing its daily dose to 1-2 g/day.
Importantly, recent clinical studies have proven the efficacy of the course of treatment with Piracetam only when administered in doses recommended by the official producer, which are 2.4-4.8 g/day [46].
WHAT ARE THE BEST RACETAMS?
Currently, there are more than 10 piracetam-like drugs, including Piracetam itself, which are already used in different countries or are at some stage of clinical trials. Not all of them are FDA approved memory supplements though.
It is important to say that the compounds of the racetam-family have a chemical structure similar to Piracetam, whose main component is a pyrrolidone ring. Despite the structural similarity, clinical effects observed in the use of racetams are quite diverse and allow to divide them into 3 subgroups: [45]
- Drugs primarily used to treat cognitive disorders. This subgroup includes Piracetam, Pramiracetam, Phenylpiracetam, Oxiracetam, and Aniracetam. Last two substances are no longer used in official clinical practice.
- Drugs for the treatment of epilepsy. This subgroup includes Levetiracetam, Brivaracetam and Seletracetam. The effect of these drugs on the cognitive function has not been established yet.
- The third subgroup includes compounds whose clinical efficacy is unknown. For example Nefiracetam (efficacy in improving cognitive function is not proven) and Rolipram (currently under study as an antidepressant). This subgroup includes a number of other drugs that are in clinical trials.
The most popular in the racetam group, except Piracetam itself, would probably be Phenylpiracetam.
NANOTROPIL (PHENYLPIRACETAM)
Similar to Piracetam, this drug has high bioavailability [26]. It was also tested a countless number of times on animals and was used in more than 500 volunteer patients before it became available to the general population.
The central neurotropic effects of Phenylpiracetam were studied and compared with Piracetam and other analogs. It was found that Phenylpiracetam has the ability to activate the operant behavior more powerfully, to remove psycho-depressant effects of diazepam, to inhibit post-rotational nystagmus, and to prevent the development of retrograde amnesia.
Unlike Piracetam, Phenylpiracetam exhibits a specific anticonvulsant action. Adding it to anticonvulsants reduces the number and frequency of seizures and slightly improves cognitive function [45].Phenylpiracetam can reduce the severity of headache and general fatigue after 14 days of therapy in patients with cognitive impairment and/or depression after TBI, and in encephalopathies of various etiology [38]. It produces a favorable effect in patients with cerebrovascular disorders [40], chronic fatigue syndrome [37], and it can help in asthenic syndrome in adolescents [41]. Phenylpiracetam is also used to increase physical and mental performance, and stimulate cognitive functions.
PRAMIRACETAM

Pramiracetam is another member of the racetam family. It is obtained by replacing the amide group in the structure of Piracetam with dipropane-2-ulaminoethyl. It allows Pramiracetam to have high bioavailability [8,17]. Pramiracetam is said to have higher potency compared to Piracetam: the most common dose is only 600mg which is 4 times fewer than that of Piracetam. Another interesting fact is that Pramiracetam has a very late onset of first effects: in most cases, they are not noted until 5-6 weeks of treatment.
Regarding the studies, Italian researchers reported a reduction in scopolamine-induced amnestic effects in healthy volunteers when using Pramiracetam, meaning that two of the five cognitive parameters including immediate and delayed verbal tests were about 50% better than in those who received placebo [24].Two studies were conducted in Ukraine: in patients with cerebrovascular disease [35] and in patients with concussion [42]. The first study showed that visual and verbal memory improved moderately with the use of Pramiracetam in younger patients with chronic cerebrovascular and poststroke cognitive symptoms and to a lesser extent in older patients.
OXIRACETAM
Due to the hydroxyl groups in the oxypyrrolidone core, Oxiracetam has a favorable pharmacokinetic profile – high bioavailability when taken orally [7].
In a prospective double-blind study in 12 healthy volunteers, Oxiracetam alleviated scopolamine-induced impairments in neuropsychiatric activity (e.g., semantic memory, reading) [20]. There is evidence that use of Oxiracetam for 2-6 months in people over 65 y.o. can improve some cognitive impairments of nonspecific etiology [21]. However, Oxiracetam failed to benefit patients with Alzheimer’s disease, although the duration of treatment was only 1 month [19].
The recommendations of the International Anti-Aging Systems (UK) state that Oxiracetam can be used for “memory impairment caused by insufficient cerebral circulation, impaired mental performance in old age”. [44] But there is no such information from the manufacturer yet.
ANIRACETAM
Due to a change in the N-side chain derivative, Aniracetam has low bioavailability and is rapidly excreted [27].
In a small study involving elderly patients with cerebrovascular disease, Aniracetam was effective [1]. However, it did not show efficiency in patients with impaired memory and cognitive functions associated with chronic toxic effects of organic solvents [14].
You can find a full Aniracetam review in the NootropicsExpert blog post.
PIRACETAM COMBINATION COMPOUNDS
Combination nootropic preparations containing Piracetam have become very popular in recent years. A synergy of nootropic and vasotropic components in one tablet/capsule seems quite reasonable [46]. The examples include piracetam and cinnarizine – Omaron, piracetam and vinpocetine – Vinpotropil, etc.
However, such drugs are usually taken when symptoms related to the proper vascular component prevail or when a combination of the effects is advisable. The symptoms include pulsation in the head, dizziness and headache of vascular origin, in some cases – excitability, anxiety, migraine. Below are some examples of such combination compounds ⬇️
VINPOTROPILE [Vinpocetine + Piracetam]
VINPOTROPILE is a combination drug. It can improve cerebral blood flow due to the active substance Vinpocetine, and it can exert a nootropic effect through Piracetam.
According to the producers the optimal dosage of the components in Vinpotropile and their proven synergy at the molecular level provide good tolerance and allow the drug to be used in a wide range of daily dosages.
According to some anecdotal experiences on Reddit it can help with tinnitus.
OMARON [Piracetam + Cinnarizine]
OMARON is another combination drug with Piracetam. It stimulates blood flow and neuronal metabolic processes in the brain and has antihypoxic, nootropic, and vasodilating effects.
One of the components is Cinnarizine. It is a beta-blocker of slow calcium channels which prevents the entry of calcium ions into cells and reduces their content in the plasma membrane. It also reduces the tone of arteriole smooth muscles and their response to biogenic vasoconstrictors such as epinephrine, norepinephrine, dopamine, angiotensin, and vasopressin. It has a vasodilating effect (especially on blood vessels in the brain) as it enhances the antihypoxic effect of Piracetam without significantly affecting arterial pressure.
In addition, the combination of Piracetam and Cinnarizine may be indicated in individual cases if a patient has a side reaction of marked excitability in response to increased doses of Piracetam as a monodrug.
PIRACETAM SOURCES
With racetam shortage it has become hard to find Piracetam online. CosmicNootropic is one reliable racetam vendor where you can buy Piracetam original and source other popular nootropics.
Piracetam at CosmicNootropic is available under the brand name Nootropil®. It is produced by UCB company which is a global biopharmaceutical leader with the headquarters in Brussels, Belgium. It specializes in the fields of central nervous system disorders, allergy and respiratory diseases, immune and inflammatory disorders, as well as oncology.
OFFICIAL INSTRUCTION – Piracetam Injections
OFFICIAL INSTRUCTION – Piracetam Pills
PIRACETAM REVIEW ON REDDIT
Piracetam has always been a popular nootropic with many positive anecdotal reviews. In a recent one a person with brain fog and memory loss was prescribed Piracetam 800mg for 2 months. Noticeable effects were reported on the 3rd week of treatment and included absence of brain fog, more energy, improved speech and general well-being.
There are also various nootropics stacks and comparisons suggested by people, for example Piracetam + Noopept or Bacopa vs Piracetam, etc.
People share different dosages and their treatment experience that includes Piracetam.
According to a statistical research shared on Reddit, Piracetam ranks 4th among the most popular top nootropics in Russia.
CONCLUSION
This review of the literature, experiences and official information on Piracetam shows that it is an enticing compound that may be of use in many brain related conditions.
Results of experimental and clinical studies accumulated to date suggest that the efficacy and safety of Piracetam and piracetam-like drugs need to be studied more deeply. The efficacy of such drugs looks promising. Therefore, there is a need for more clinical trials.
It shall also be pointed out that Piracetam and piracetam-like drugs are relatively safe. However, it can be said that the delayed effects of these drugs and the potential risks associated with their use have not been identified to the fullest yet. The mechanisms of action of Piracetam and piracetam-like drugs are also understood insufficiently and require further clarification. But the prospects for the use of Piracetam in clinical practice are in no doubt.
PIRACETAM REVIEW REFERENCES
- Buresová O, Bures J (1976). Piracetam-induced facilitation of interhemispheric transfer of visual information in rats. https://pubmed.ncbi.nlm.nih.gov/1257371/
- Abuzzahab FS et al (1977). A double blind investigation of piracetam (Nootropil) vs placebo in geriatric memory. https://pubmed.ncbi.nlm.nih.gov/360232/
- Valzelli L, Bernasconi S, Sala A (1980). Piracetam activity may differ according to the age of the recipient mouse. https://pubmed.ncbi.nlm.nih.gov/6165698/
- Wurtman RJ, Magic SG, Reinstein DK (1981). Piracetam diminishes hippocampal acetylcholine levels in rats. https://pubmed.ncbi.nlm.nih.gov/7231039/
- Olpe H-R, Steinmann MW (1981). Rapid communication the activating activity of vincamine, piracetam, and hydergine on the activity of the noradrenergic neurons of the locus coeruleus. https://pubmed.ncbi.nlm.nih.gov/7305817/
- Bartus RT et al (1981). Profound effects of combining choline and piracetam on memory enhancement and cholinergic function in aged rats. https://pubmed.ncbi.nlm.nih.gov/7301036/
- Perucca E, Albrici A, Gatti G, et al (1984). Pharmacokinetics of oxiracetam following intravenous and oral administration in healthy volunteers. https://link.springer.com/article/10.1007/BF03189650
- Chang T, Young RM, Goulet JR, et al (1985). Pharmacokinetics of oral pramiracetam in normal volunteers. https://pubmed.ncbi.nlm.nih.gov/4008675/
- Herrmann WM, Kern U (1987). Nootropic drugs – effects and therapeutic efficacy: A phase III study with piracetam as a model. https://pubmed.ncbi.nlm.nih.gov/3302738/
- Wilsher CR et al (1987). Piracetam and dyslexia: effects on reading tests. https://pubmed.ncbi.nlm.nih.gov/3305591/
- Pilch H, Müller WE (1988). Piracetam elevates muscarinic cholinergic receptor density in the frontal cortex of aged but not of young mice. https://pubmed.ncbi.nlm.nih.gov/3126530/
- Mondadori C, Ducret T, Borkowski J (1989). The memory-enhancing effects of the piracetam-like nootropics are dependent on experimental parameters. https://pubmed.ncbi.nlm.nih.gov/2736062/
- Voronina TA (1989). Pharmacology of Nootropics. https://search.rsl.ru/ru/record/01001477472
- Somnier FE, Ostergaard MS, Boysen G, et al (1990). Aniracetam tested in chronic psychosyndrome after long-term exposure to organic solvents. A randomized, double-blind, placebo-controlled cross-over study with neuropsychological tests. https://pubmed.ncbi.nlm.nih.gov/2188276/
- Fioravanti M, et al (1991). A multicentre, double-blind, controlled study of Piracetam vs placebo in geriatric patients with nonvascular mild-moderate impairment in cognition. https://moh-it.pure.elsevier.com/en/publications/a-multicentre-double-blind-controlled-study-of-piracetam-vs-place
- Vernon MW, Sorkin EM (1991). Piracetam. An overview of its pharmacological properties and a review of its therapeutic use in senile cognitive disorders. https://pubmed.ncbi.nlm.nih.gov/1794001/
- Auteri A, Blardi P, Celasco G, et al (1992). Pharmacokinetics of pramiracetam in healthy volunteers after oral administration. https://pubmed.ncbi.nlm.nih.gov/1473879/
- Stoll L, Schubert T, Müller WE (1992). Age-related deficits of central muscarinic cholinergic receptor function in the mouse: partial restoration by chronic piracetam treatment. https://pubmed.ncbi.nlm.nih.gov/1542379/
- Green RC, Goldstein FC, Auchus AP, et al (1992). Treatment trial of oxiracetam in Alzheimer’s disease. https://pubmed.ncbi.nlm.nih.gov/1444879/
- Preda L, Alberoni M, Bressi S, et al (1993). Effects of acute doses of oxiracetam in the scopolamine model of human amnesia. https://pubmed.ncbi.nlm.nih.gov/7870912/
- Rozzini R, Zanetti O, Bianchetti A (1993). Treatment of cognitive impairment secondary to degenerative dementia: effectiveness of oxiracetam therapy. https://pubmed.ncbi.nlm.nih.gov/8456595/
- Croisile B et al (1993). Long-term and high-dose piracetam treatment of Alzheimer’s disease. https://pubmed.ncbi.nlm.nih.gov/8437693/
- Enderby P et al (1994). Effect of piracetam on recovery and rehabilitation after stroke: a double-blind, placebo-controlled study. https://pubmed.ncbi.nlm.nih.gov/9316679/
- Mauri M, Sinforiani E, Reverberi F, et al (1994). Pramiracetam effects on scopolamine–induced amnesia in healthy volunteers. https://pubmed.ncbi.nlm.nih.gov/15374306/
- Gouliaev AH, Senning A (1994). Piracetam and other structurally related nootropics. https://pubmed.ncbi.nlm.nih.gov/8061686/
- Spektor SS, Berlyand AS (1996). Molecular–biological problems of drug design and mechanisms of drug action: experimental pharmacokinetics of carphedon. https://link.springer.com/article/10.1007/BF02334630
- Ogiso T, Iwaki M, Tanino T, et al (2000). Pharmacokinetics of aniracetam and its metabolites in rat brain. https://pubmed.ncbi.nlm.nih.gov/10784432/
- Avedisova AS, Ahapkin RV, Ahapkina VI, Verigo NN (2000). Piracetam in the light of modern research (analysis of foreign studies). http://bono-esse.ru/blizzard/Farma/Pyracetam.pdf
- Segonia G, Porras A, Del Arco A, Mora F (2001). Glutamatergic neurotransmission in aging: A critical perspective. https://pubmed.ncbi.nlm.nih.gov/11163621/
- Gualtien F, Manetti D, Romanelli MN, Ghelardini C (2002). Design and study of piracetam-like nootropics, controversial members of the problematic class of cognition-enhancing drugs. https://pubmed.ncbi.nlm.nih.gov/11812254/
- Avedisova AS, Verigo NN, Brutman VI (2002). Comparative analysis of high and usual doses of Lucetam (piracetam) in patients with residual schizophrenia. https://www-rmj-ru.translate.goog/articles/obshchie-stati/sravnitelynyy_analiz_primeneniya_vysokih_i_obychnyh_doz_lucetama_piracetama_u_bolynyh_rezidualynoy_shizofreniey/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Waegemans T, Wilsher CR, et al. (2002). Clinical efficacy of piracetam in cognitive impairment: a meta-analysis. https://pubmed.ncbi.nlm.nih.gov/12006732/
- Parfenov VA (2002). Metabolic therapy for ischemic stroke. https://www-rmj-ru.translate.goog/articles/nevrologiya/Metabolicheskaya_terapiya_ishemicheskogo_insulyta/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Aleksandrovskij YuA (2003). Psychiatry and Psychopharmacotherapy. Selected Lectures and Speeches. https://www.studmed.ru/view/aleksandrovskiy-yua-psihofarmakoterapiya_2b581ce666f.html
- Dziak LA, Golik VA, Miziakina EV (2003). Experience in the application of pramistar, a new nootropic preparation, in the treatment of memory disorders in patients with cerebrovascular pathology. https://pubmed.ncbi.nlm.nih.gov/14965012/
- Terry AVJr, Buccafusco JJ (2003). The cholinergic hypothesis of age and Alzheimer’s disease-related cognitive deficits: recent challenges and their implications for novel drug development. https://pubmed.ncbi.nlm.nih.gov/12805474/
- Akhapkina VI et al (2004). Phenotropil efficacy in the treatment of asthenic syndrome and chronic fatigue syndrome. https://medi-ru.translate.goog/info/9517/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Savchenko AYu, Zakharova NS, Stepanov IN (2005). The phenotropil treatment of the consequences of brain organic lesions. https://medi-ru.translate.goog/info/11676/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Winblad B (2005). Piracetam: a review of pharmacological properties and clinical uses. https://onlinelibrary.wiley.com/doi/pdf/10.1111/j.1527-3458.2005.tb00268.x
- Gustov AA et al (2006). Phenotropil in the treatment of vascular encephalopathy. https://medi-ru.translate.goog/info/3831/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Zvonareva EV (2006). Phenotropil in the therapy of cognitive disorders in teenagers with asthenic syndrome. https://cyberleninka.ru/article/n/fenotropil-v-terapii-kognitivnyh-rasstroystv-u-podrostkov-s-astenicheskim-sindromom
- Tkachev AV (2007). Application of nootropic agents in complex treatment of patients with concussion of the brain. https://pubmed.ncbi.nlm.nih.gov/18418926/
- Damulin IV (2008). Vascular dementia: some pathogenetic, diagnostic and therapeutic aspects. https://www-rmj-ru.translate.goog/articles/nevrologiya/Sosudistaya_demenciya_nekotorye_patogeneticheskie_diagnosticheskie_i_terapevticheskie_aspekty/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- Malykh AG, Sadaie MR (2010). Piracetam and piracetam-like drugs: from basic science to novel clinical applications to CNS disorders. https://pubmed.ncbi.nlm.nih.gov/20166767/
- Sychev DA et al (2011). Piracetam and piracetam-like drugs: a clinical pharmacologist’s perspective. https://www-rmj-ru.translate.goog/articles/nevrologiya/Piracetam_i_piracetamopodobnye_preparaty_vzglyad_klinicheskogo_farmakologa/?_x_tr_sl=ru&_x_tr_tl=en&_x_tr_hl=ru&_x_tr_pto=wapp
- V.V. Vostrikov (2017). Place Of Piracetam In The Modern Practice Of Medicine. https://www.researchgate.net/publication/316174250_Place_of_piracetam_in_the_modern_practice_of_medicine
- Zongfang Z et al (2020). Therapeutic effect of piracetam with nimodipine on vascular dementia after cerebral infarction. https://pubmed.ncbi.nlm.nih.gov/33832882/






